One would think it would be easier to concoct artificial blood in laboratories (given that we know all its components) than to fabricate the intangible intelligence. But here we are, with AI ready to take over the world, still wondering-
Why we cannot create Artificial blood?
Life in circulation: BLOOD- Composite Fluid vs. Dynamic organ system
It would be imprudent to consider blood as yet another body fluid/secretion. With multiple, tightly regulated cell lineages, that carry identifications unique to individual members of each species, blood is a highly specialised connective tissue on the move with every heartbeat.
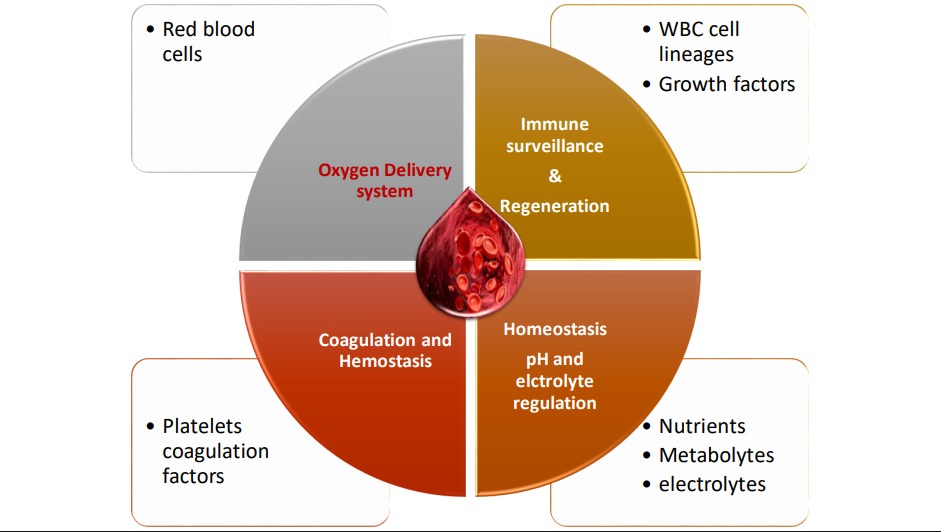
While blood in circulation is primarily considered to be the most efficient oxygen delivery system, it functions as a carrier of nutrients, waste products, immune cells and factors, regenerative factors, and coagulation proteins.
Graphic ©Dr. Sai Rane 2025
Naturally a completely functional, ideal, artificial blood substitute would be expected to carry most of these functions as well as respond to internal changes and external threats.
About 55% blood comprises of fluid but coagulable plasma, consisting of soluble proteins like albumin, globulins, complement factors, enzymes, hormones and signalling molecules that respond to various immunological, metabolic, and hormonal stimuli.
The remaining 45% blood comprises of cells, in which 44% are made up by the red blood cells responsible for gaseous exchange at the cellular level; and the white blood cells and platelets making up the last 1%.
It is this last 1% that is hardest to duplicate, as it is unique to each individual at any given time, and differentiates you from anyone that is not you. These highly labile cells of the adaptive immune system, carrying signature markers based on infectious exposures, that vary from time to time even within a single individual, pose the biggest challenge in artificial fabrication of functional whole blood (with adaptive immune function). Current artificial blood substitutes are therefore inherently immunologically inert. Any attempt to synthesise immunologically functional blood substitute would thus involve a reconstruction of a viable hematopoietic system.
Similarly, the intricate cascades of haemostatic mechanisms orchestrated by the platelets and the complement system require a certain local intelligence that enables it to remain inert/ inactive in normal circulation, but respond promptly to capillary injury (tissue factor expression), and must also possess self-regulatory mechanisms to terminate the cascade once haemostasis is achieved, in order to prevent excessive clotting. Duplicating these elaborate feedback loops using lab made inert molecules is currently theoretically unattainable.
Artificial Blood/ Blood substitutes through history
Through centuries, medicine men have been in search of ways to prevent fatalities due to severe blood loss. Going by the historic records various solutions were known to be used:
– old blood transfusions (which were often unsuccessful due to ignorance of the existence of blood types),
– milk (yes, you read it right! Milk was touted to be a ‘brilliant’ substitute by 19th century gynaecologist, Theodore Gaillard Thomas),
– saline solutions (still in use today).
– volume expanders
We have now gone back to whole blood transfusions using carefully perfected cross matching techniques which remains the gold standard.
THE DEMAND
According to a recent cross sectional study (Mammen et al, 2022[1]), that evaluated 251 health facilities with 51,562 beds in India, the total clinical demand was 474,627 whole blood units- which can be extrapolated to an estimated clinical demand of 14·6 million whole blood units for our nation This translates to an equivalent of 36·3 donations per 1,000 eligible populations, to address whole blood and component requirement. The supply was 93% which is equivalent to 33·8 donations against the demand— reflecting a shortage of 2.5 donations per 1000 populations.
LIMITATIONS OF WHOLE BLOOD TRANSFUSIONS
- Limited supply for increasing demand
- Shortage of facilities with ideal storage specifications
- Errors in cross-matching
- Rejection of transfused blood due to multiple factors.
THE REALM OF BIOENGINEERING ARTIFICIAL BLOOD: Holy grail of Biomedical research
Dr. Keith Neeves of Haemophilia and Thrombosis Centre of University of Colorado School of Medicine, describes of two possible approaches to attain goal of synthesis of artificial blood.
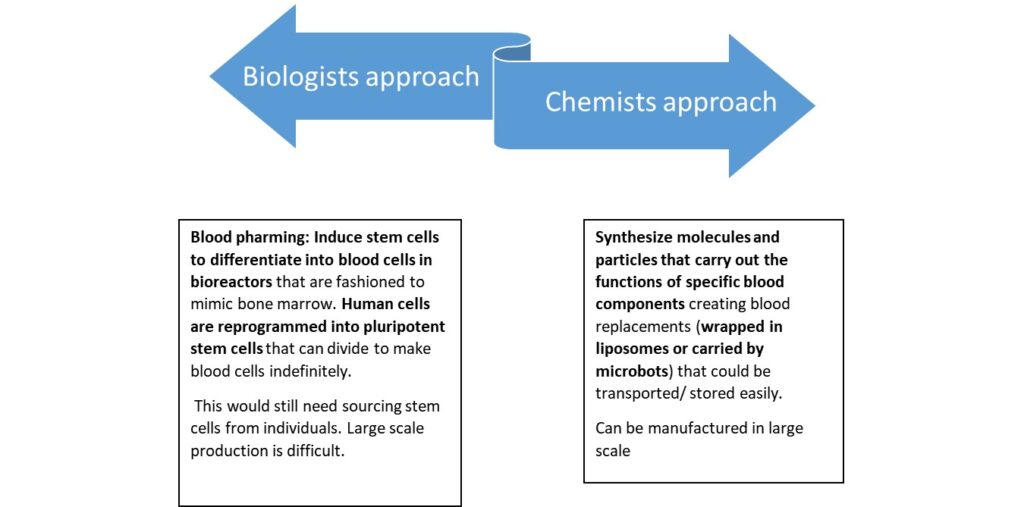
CHEMISTS APPROACH- SYNTHETIC BLOOD:
Existing research efforts essentially focus almost exclusively on blood substitutes, which focus on tiding over the acute hypoxic and hypovolemic stat, providing a temporary bridge until natural blood products can be administered, and are hence more appropriately termed Oxygen Therapeutic Agents (OTAs). They do not, and currently cannot, replace the immunological, homeostatic, or sustained clotting capabilities of natural blood.
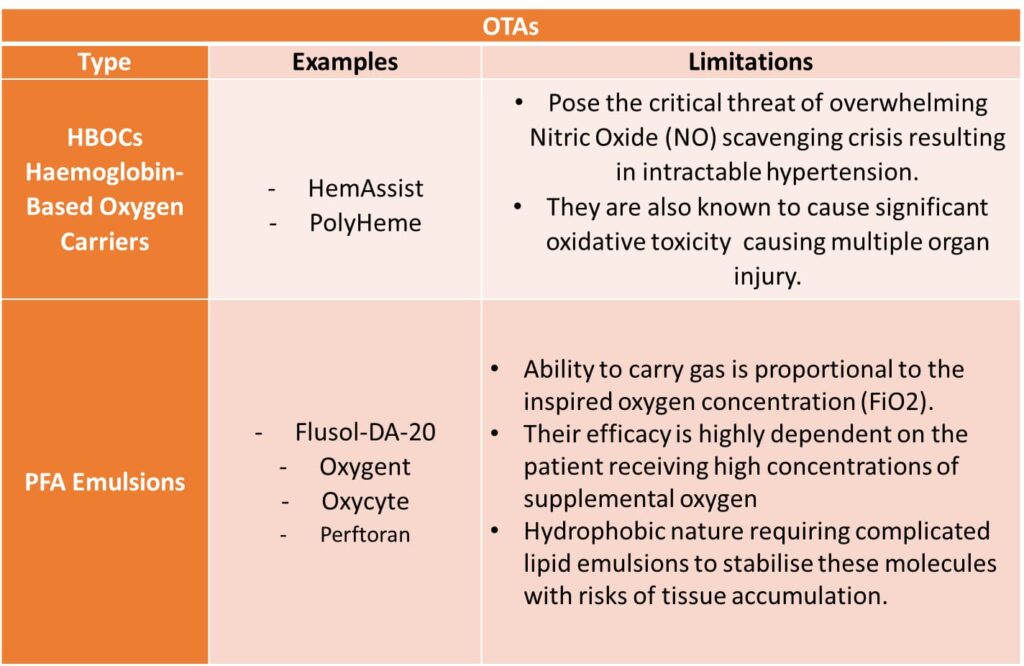
Currently researched OTAs include:
- Haemoglobin-Based Oxygen Carriers (HBOCs)
- Perfluorocarbon-based oxygen carriers (PBOC/ PFC Emulsions)
Challenges in replicating oxygen transport dynamics:
The tiny RBC is not a simplistic bag carrying haemoglobin but a far more complex biochemical vessel that can intelligently regulate its contents. Haemoglobin, the incredible oxygen carrying molecule is inherently toxic (nephrotoxic in elevated doses) when it exists as a cell-free molecule floating in plasma. This emphasizes the critical role played by the RBC membrane to efficiently contains this protein, prevent its rapid clearance and offer immunologic protection.
Chemically modified Haemoglobin contained in synthetic cell membranes (liposomes) are being researched to bypass the toxicity and immunologic issues, but achieving dynamic control over the binding and release of oxygen molecules in high and low oxygen concentration milieu, remains challenging.
This intelligent dynamic control over release of oxygen in deoxygenated tissues is regulated by a simple molecule known as 2,3-Bisphosphoglycerate (2,3-BPG), which reduces the affinity of haemoglobin to oxygen allowing its unloading in the tissues (shift to right of oxygen dissociation curve).
Currently none of the synthetic or partially synthetic carriers carry this ability sense the tissue demand and synthesize this molecule to modulate oxygen affinity resulting in inefficient oxygenation of body tissues.
While PFC emulsions a promising in that they can be produced in mass scales, they are by nature hydrophobic and require specialised lipophilic carriers that also carry a risk of accumulating in the tissues.
These shortcomings apart OTAs offer certain promises:
- They have significantly extended shelf lives (up to two years at ambient temperature)
- They are universally compatible, eliminating the need for blood typing and refrigeration.
These characteristics make them highly valuable for specific applications, particularly in military, rural, or pre-hospital trauma settings where delays in obtaining refrigerated, typed donor blood can be fatal.
BLOOD PHARMING:
This innovative approach involves manufacturing red blood cells (mRBCs) from haematopoietic cell lineages or induced Pluripotent Stem Cells (iPSCs), erythroblast progenitor cells and immortalised progenitor cell lines, directing them towards erythropoiesis in an in vitro environment.
The “RESTORE (Recovery and Survival of Stem Cell Originated Red Cells) trial”, an initiative by the “NHS Blood and Transplant, University of Bristol, National Institute for Health and Care Research Cambridge Clinical Facility” first reported a breakthrough in the field of transfusion medicine. The trial was aimed to compare the post transfusion longevity of mRBCs which were developed from donor−derived stem cells compared to the standard RBC transfusion from the same donor.
The scalability challenges of blood pharming are currently unsurmountable. For perspective- production of a single unit of transfused blood contains a massive quantity of cells, approximately 1-2 x 1012 red cells. Based on current maximum densities achievable in static flask cultures (roughly 2 x 106 cells/ml), producing just one unit of blood would require over 1,000 litres of culture medium translating into engineering difficulties of monumental proportions to meet national demand levels.
Then there are the ethical and regulatory roadblocks in researching artificially engineered blood products from stem cells. Haemodynamically deficient individuals are hardly ever in a condition to give an informed consent.
Future strategies and research priorities
High functional blood substitute production requires solving the dichotomy of between achieving biological, cell-based systems (stem cells), and mass production requiring economical, chemical synthesis.
Targeted component replacement: Based on the cause of volume depletion, availability of assorted artificial blood components may offer flexible solutions and appears to be the most achievable goal. Reliable shelf stable components like artificial platelet analogues, specific coagulation proteins that can be used to supplement existing banked blood products or plasma.
Scalable biologic production of selected cell lines: For specific needs such as rare blood types or specific immune cell components.
Countering or eliminating toxic potential of chemical replacement products: Encapsulation of artificial Haemoglobin proteins with advanced liposome technologies with an attempt to attain dynamism in oxygen binding properties.
It is just about five litres of life circulating through our veins, (bizarrely made up by 32 trillion of the total 36 trillion cells that make us), being replenished and purified, over and over, keeping us alive until for some reason, it stops.
And while we have been successful in creating artificial intelligence (which, by the way, can give an incredibly neat summary on why artificial blood can’t be created) we haven’t yet been able to concoct this magical fluid artificially in our ultramodern laboratories. We still depend on mother nature and her progenies for its production.
References:
- Mammen JJ, Asirvatham ES, Lakshmanan J, Sarman CJ, Pandey A, Ranjan V, Charles B, Mani T, Khaparde SD, Upadhyaya S, Rajan S. The clinical demand and supply of blood in India: A National level estimation study. PLoS One. 2022 Apr 6;17(4)
https://pmc.ncbi.nlm.nih.gov/articles/PMC8986005/
2) Mark Harden | November 15, 2024 | There’s a Shortage of Blood. So Why Don’t We Make Some? https://news.cuanschutz.edu/medicine/neeves-synthetic-blood-bioengineering
3) Dr. Keith Neeves Jul 14, 2024 Where’s the Synthetic Blood? https://www.asimov.press/p/synthetic-blood
4) Why is artificial blood so hard to make? | Science – The Guardian, https://www.theguardian.com/science/2004/may/20/thisweekssciencequestions
5) Manxu Zhao, Henry Liu, Jonathan S Jahr, Perfluorocarbon-based oxygen carriers: What is new in 2024?,Journal of Anesthesia and Translational Medicine,Volume 3, Issue 1, 2024 Perfluorocarbon-based oxygen carriers: What is new in 2024? – ScienceDirect
6) Blood pharming: exploring the progress and hurdles in producing in-vitro red blood cells for therapeutic applications Front. Hematol., 28 March 2024 Sec. Hematopoiesis and Stem Cells Volume 3 2024| https://doi.org/10.3389/frhem.2024.1373408
7) Why is artificial blood so hard to make? | Science – The Guardian, https://www.theguardian.com/science/2004/may/20/thisweekssciencequestions
8) Manxu Zhao, Henry Liu, Jonathan S Jahr, Perfluorocarbon-based oxygen carriers: What is new in 2024?,Journal of Anesthesia and Translational Medicine,Volume 3, Issue 1, 2024 Perfluorocarbon-based oxygen carriers: What is new in 2024? – ScienceDirect
9) Blood pharming: exploring the progress and hurdles in producing in-vitro red blood cells for therapeutic applications Front. Hematol., 28 March 2024 Sec. Hematopoiesis and Stem Cells Volume 3 2024| https://doi.org/10.3389/frhem.2024.1373408\